What is Benedict's Reagent?
 Tricia Christensen
Tricia Christensen
Benedict’s reagent is a solution of copper sulfate, sodium carbonate and sodium citrate in water. It is used to detect the presence of certain types of carbohydrate known as reducing sugars. These substances can undergo chemical reactions in which they give electrons to other compounds, resulting in the production of new substances, and they react in this way with Benedict’s reagent to produce an insoluble, reddish colored compound. Glucose and fructose produce a positive reaction, but sucrose — table sugar — does not. The reagent is used in food testing, and to detect glucose in urine, which can be a sign of diabetes.
Types of Carbohydrate

Carbohydrates can be monosaccharides, which are simple molecules such as glucose (C6H12O6); disaccharides, which consist of two monosaccharides bonded together, for example sucrose; or polysaccharides, which are long chains of many monosaccharide units. Monosaccharides always have a carbonyl group — a carbon atom joined to an oxygen atom by a double bond — that reacts with Benedict’s reagent. Some disaccharides, such as maltose and lactose, have carbonyl groups, and some do not; it depends upon how the monosaccharide units are joined together. In sucrose, a glucose and a fructose molecule are joined in such a way that their carbonyl groups are broken. Polysaccharides, such as starch, have very few of these groups, and so produce little or no reaction.
How it Works

Reducing sugars react with the copper sulfate in Benedict’s reagent, reducing it to copper I oxide, an insoluble, reddish colored compound that forms a precipitate. The sodium carbonate is required to make the solution alkaline, which is essential to allow some types of carbohydrate to react, while the sodium citrate prevents the copper sulfate from reacting with the alkali. The solution is blue in color, due to the copper sulfate. The test is essentially qualitative, that is, it is used simply to check whether or not a reducing sugar is present, and not to determine the quantity. It can, however, be used as a crude quantitative test, in that a greenish color indicates only a little reducing sugar; yellow, a bit more; and red, a lot.

Another reagent, known as quantitative Benedict’s solution (QBS), can be used to determine, quite precisely, how much reducing sugar is present in a sample. It is similar to the normal reagent, but contains two additional chemicals. In this solution, a positive result is indicated by a white precipitate and loss of some of the initial blue color. The intensity of the color indicates the quantity of reducing sugar in the sample, and can be measured using a device called a colorimeter.
Use in Food Testing

Foods can be tested for reducing sugars by mashing or grinding a small amount and adding it to some Benedict’s reagent in a test tube, then heating for several minutes. The color of the resulting solution indicates whether any of these compounds are present and gives a rough idea of how much. This test will detect sugars commonly present in food, such as glucose, fructose, maltose and lactose. It will not, however, detect sucrose, which is the type most commonly added to processed foods. Boiling sucrose with dilute hydrochloric acid will split it into glucose and fructose, which can then be detected.
Use in Medicine
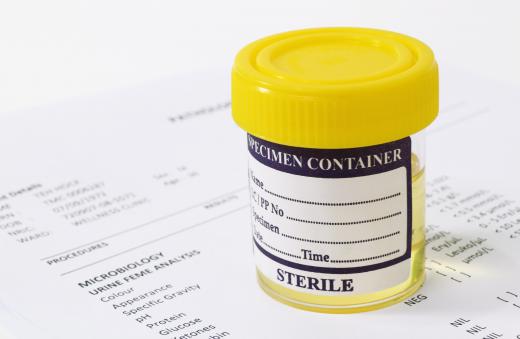
The presence of glucose in urine can be a sign of diabetes. Testing a urine sample with Benedict's reagent is a simple way of checking for the presence of glucose in people who are suspected of having this disease. It is, however, not a definitive test, as other reducing sugars will produce the same reaction. If urine tests positive, further tests will have to be carried out to confirm the condition. Pregnant women may be tested in this way at regular intervals to detect gestational diabetes, which can appear during pregnancy in women with no previous history of the condition.
AS FEATURED ON:
AS FEATURED ON:













Discussion Comments
What colours indicate diabetes?
I was just wondering if you could tell me why it would be important to measure the concentration of glucose in a solution before conducting the experiment.
Give the reaction equation of benedict solution with sugars.
How would a crude yeast homogenate be used where only sucrase is tested?
What is the actual cause of the color change beyond it just being due to the carbohydrate present?
What is the chemical makeup?
What colors can it turn?
is there any difference between this and the Somogyi Reagent?
In terms of its structure, why is sucrose a non reducing sugar?
what are the tests that are used to evaluate for glucose only (since Benedict's reagent can be used for both glucose and fructose)?
How do we test for different amount of reducing sugar in different samples. It is the different amount of precipitate that forms that indicates how much reducing sugar is present, but how can we record down in a report?
Are there any other functions of Benedict's Reagent?
Post your comments